 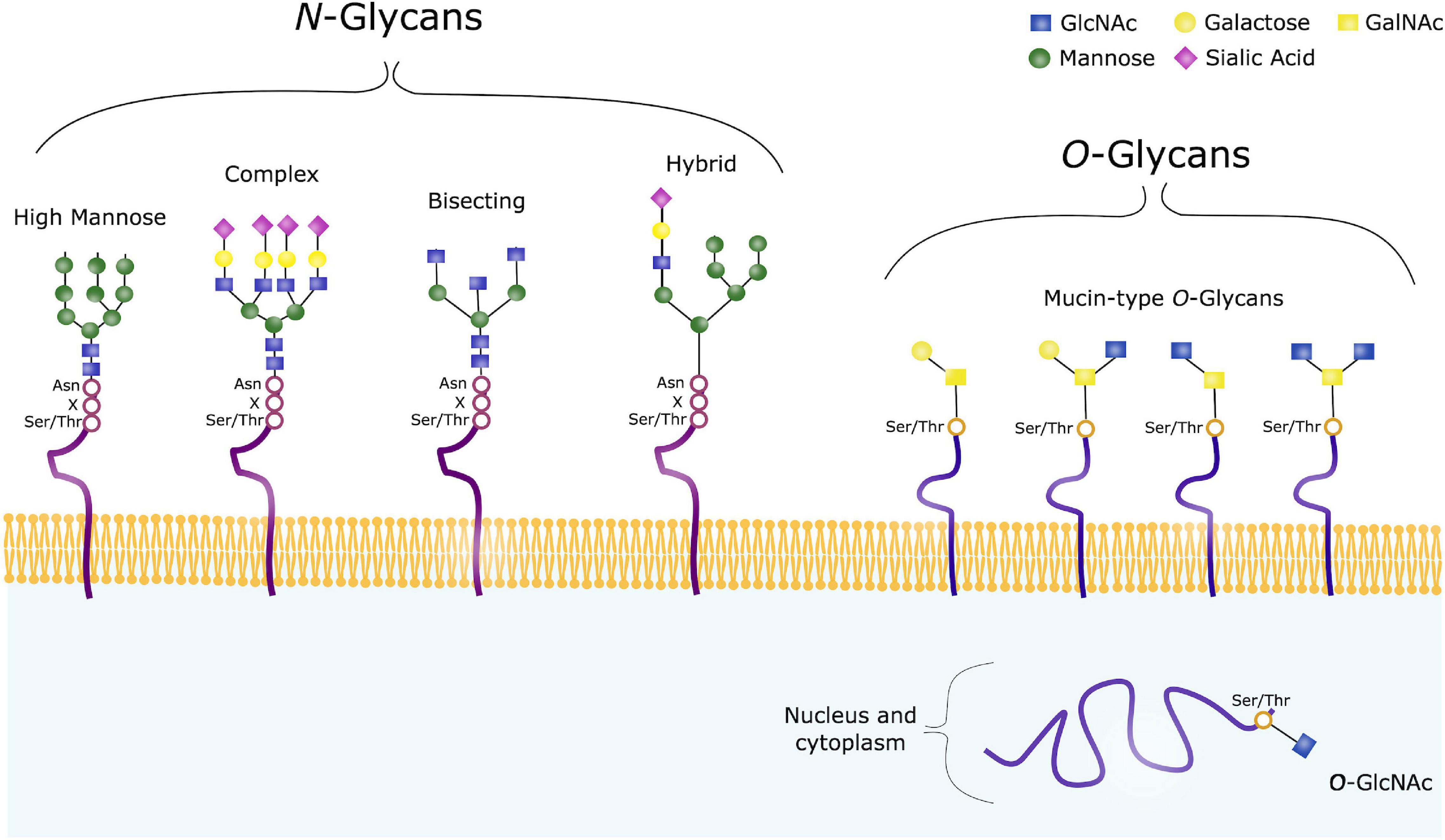
Core 2 consists of a Core 1 structure with an additional N-acetylglucosamine (GlcNAc) sugar. Core 1 is formed by the addition of a galactose sugar onto the initial GalNAc. Two of the most common structures formed are Core 1 and Core 2. Once this initial sugar has been added, other glycosyltransferases can catalyse the addition of additional sugars. However, there are often proline (Pro) residues near the threonine or serine. The specific residue onto which GalNAc will be attached is not defined, because there are numerous enzymes that can add the sugar and each one will favour different residues. This precursor is necessary so that the sugar can be transported to where it will be added to the protein. GalNAc is added onto a serine or threonine residue from a precursor molecule, through the activity of a GalNAc transferase enzyme. Galactose (Gal) can be added to form the B-antigen. N-acetylgalactosamine (GalNAc) can be added to the H-antigen to form the A-antigen. O-glycosylation occurs in all domains of life, including eukaryotes, archaea and a number of pathogenic bacteria including Burkholderia cenocepacia, Neisseria gonorrhoeae and Acinetobacter baumannii. Because of the many functions they have, changes in O-glycosylation are important in many diseases including cancer, diabetes and Alzheimer's. O-glycans, which are the sugars added to the serine or threonine, have numerous functions throughout the body, including trafficking of cells in the immune system, allowing recognition of foreign material, controlling cell metabolism and providing cartilage and tendon flexibility. Several different sugars can be added to the serine or threonine, and they affect the protein in different ways by changing protein stability and regulating protein activity. In eukaryotes, it occurs in the endoplasmic reticulum, Golgi apparatus and occasionally in the cytoplasm in prokaryotes, it occurs in the cytoplasm. O-glycosylation is a post-translational modification that occurs after the protein has been synthesised. 
O-linked glycosylation is the attachment of a sugar molecule to the oxygen atom of serine (Ser) or threonine (Thr) residues in a protein. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
